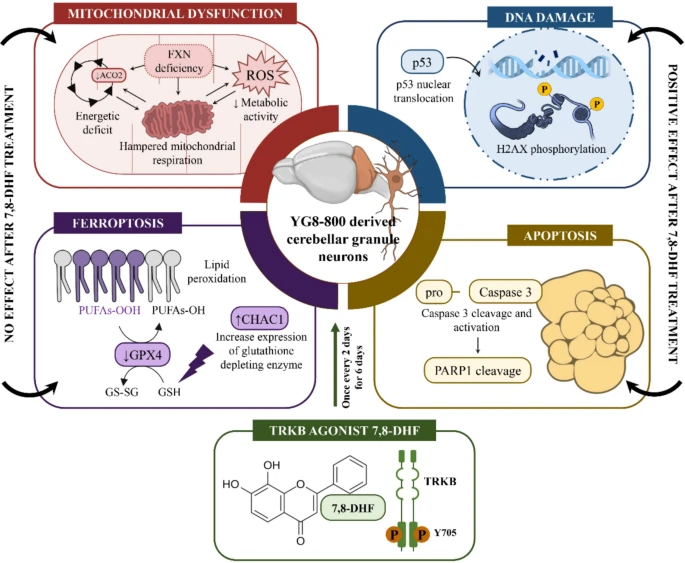
We found evidence of mitochondrial dysfunction concomitant with DNA damage and enhanced cell death due to FXN deficiency in cultured neurons. The treatment with 7,8-DHF was able to reduce the markers of genotoxicity and apoptosis, without restoring the impaired mitochondrial function nor the total cell death, possibly through ferroptosis, revealing a partial neuroprotective effect insufficient to halt the neurodegenerative process in this in vitro model of FRDA.
Monday, April 27, 2026
The TRKB Agonist 7,8-dihydroxyflavone Alleviates DNA Damage and Apoptosis in a Neuronal Cell Model of Friedreich’s Ataxia
Galán-Cruz, J., Vicente-Acosta, A., Loría, F. et al. The TRKB Agonist 7,8-dihydroxyflavone Alleviates DNA Damage and Apoptosis in a Neuronal Cell Model of Friedreich’s Ataxia. Mol Neurobiol 63, 580 (2026). doi:10.1007/s12035-026-05856-2
Novel Precision Gene Therapy for Friedreich’s Ataxia with Dr. Gabriel Brooks Solid Biosciences
Apr 23, 2026 . Solid Biosciences
Dr. Gabriel Brooks is Chief Medical Officer at Solid Biosciences, a precision molecular genetic medicines company focused on rare cardiovascular and neuromuscular diseases, including Friedreich's ataxia. Currently, there are very limited treatments for this rare, progressive neurologic disease caused by a genetic deficiency. Solid Biowsciences' novel gene therapy uses dual-route administration to deliver directly to the heart and brain and to replace the missing frataxin gene, which is critical for energy production.
Nomlabofusp Treatment Produces Frataxin Levels That Correlate Across Peripheral Tissues: Preclinical and Clinical Support for Surrogate Tissue Sampling
De Toni F, Hamdani M, Patadia C, Shankar G. Nomlabofusp Treatment Produces Frataxin Levels That Correlate Across Peripheral Tissues: Preclinical and Clinical Support for Surrogate Tissue Sampling. Clin Transl Sci. 2026 May;19(5):e70565. doi: 10.1111/cts.70565. PMID: 42033121; PMCID: PMC13109711.
This study evaluated whether nomlabofusp‐derived hFXN concentrations covary across accessible peripheral matrices and FRDA‐relevant tissues, supporting the feasibility of surrogate tissue sampling to monitor drug‐derived hFXN exposure.
Subscribe to:
Posts (Atom)